a shiny solid element also is ductile. what side of the periodic table is it likely to be found
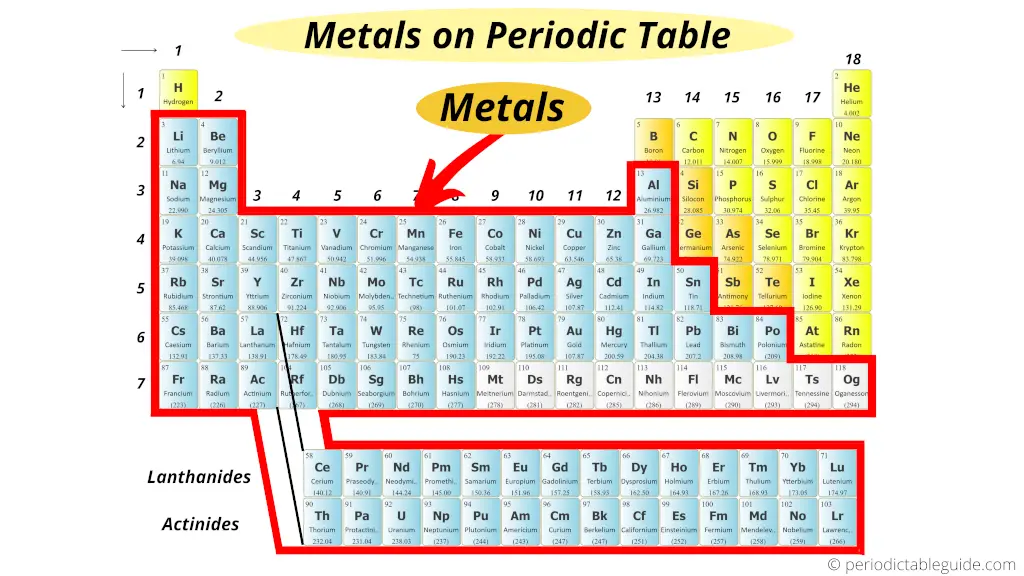
The metals are located on the left side of the Periodic Tabular array.
This is the short and elementary reply of your question "Where are Metals located on the Periodic Table?"
But wait…
There are lot more than things yous need to know nearly the metals of the Periodic tabular array, like;
- Alkali metals on Periodic table
- Element of group i World metals on Periodic table
- Transition metals on Periodic table
- Inner transition metals on Periodic table
- How many metals are at that place on Periodic table?
- Rare Earth metals on Periodic table
- Heavy metals on Periodic tabular array
- Reactive metals on Periodic table
- The complete list of all metals
- Why are metals on the left of Periodic tabular array?
- Properties of metals
- Physical properties of metals
- Chemical properties of metals
- And lots more…
Allow's dive straight into it.
What are metals in Periodic table?
Metals are the elements which have the trend to donate or lose electrons to course positive ions.
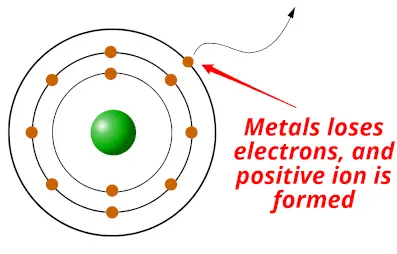
In brusk remember that;
- Metals are electron donors (metals loses / donates electrons)
The elements which show this type of nature (nature of losing electrons) are known as metals.
Now in the Periodic tabular array, these metals are found on the left side.
Just these metals are further classified into many more types.
Permit me tell yous few things well-nigh the types of metals on the Periodic tabular array.
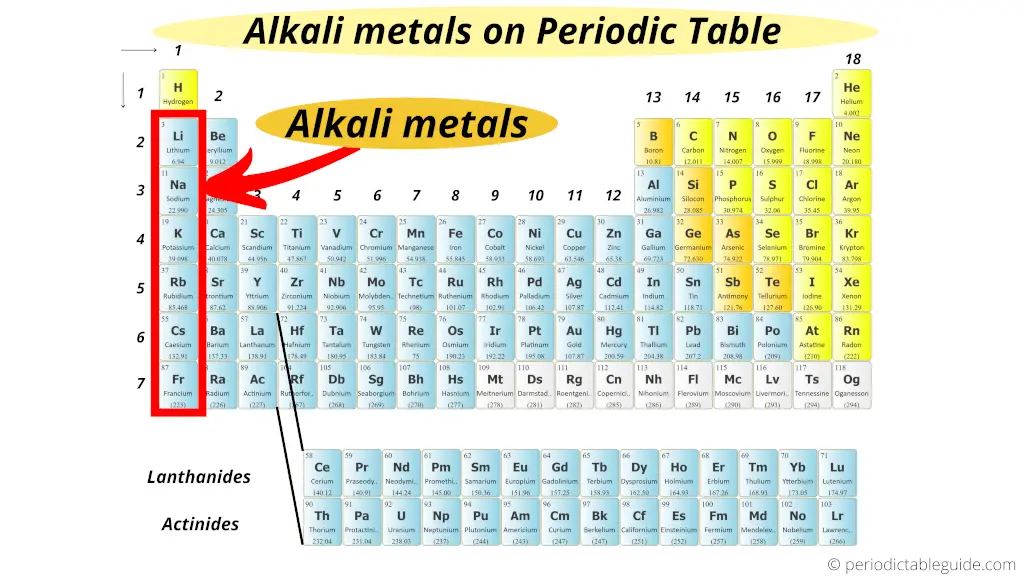
The left near elements on the Periodic table (group 1 elements) are known every bit alkali metals.
Simply practice you know;
What are alkali metals? And why the alkali metals are called so?
The elementary reply: Alkali metals form an alkaline metal solution (basic solution) when they react with h2o.
That's why they are known as alkali metals. (Read detailed explanation from here.)
Likewise visit: Why are brine metals and so reactive?
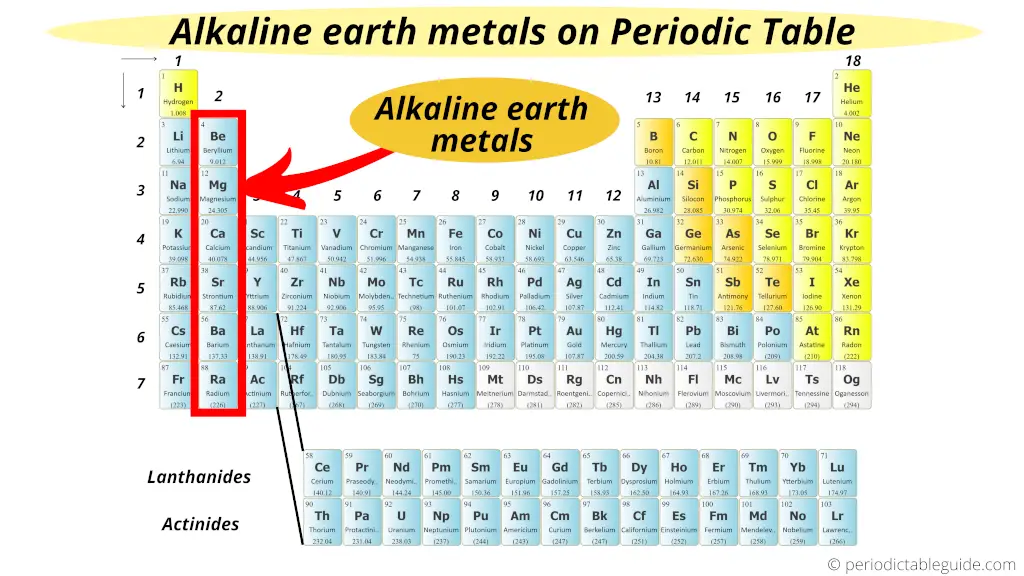
The Alkaline Earth metals are located in the 2d group of Periodic table.
Now again I will tell you a simple reason why these group 2 elements are known every bit Element of group i Earth Metals.
Encounter, the grouping 2 elements take almost similar properties that of the grouping 1 elements (Alkali Metals).
But the difference is that they are mostly institute from the world crust.
Thus they are known as Alkali metal World Metals.
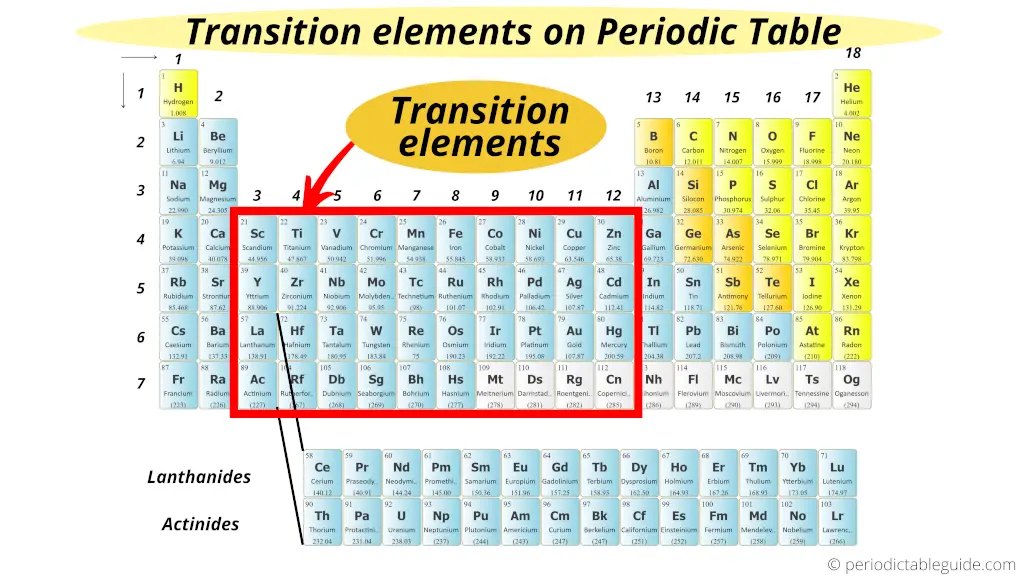
The elements lying in group 3 to group 12 are known every bit Transition metals (or transition elements).
Transition metals form a span between the chemically active metals of s-cake elements and the less agile elements of Groups xiii and 14.
Thus these metals are known every bit "Transition metals".
Also visit: How many transition metals are there on periodic table?
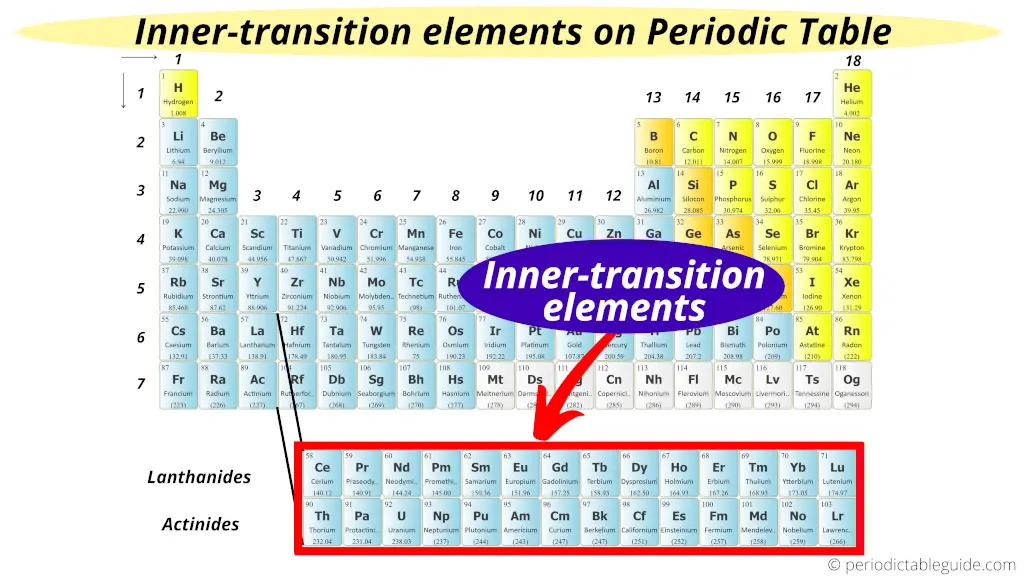
The two bottom rows in the Periodic table are called inner transition metals.
Why?
Just equally the name suggests, they are transition metals only, but they are lying in the inner section of the transition metals.
Hence they are named every bit inner transition metals (or inner transition elements)
The elements of the beginning row are known as Lanthanides and the elements of the second row are known as Actinides.
Likewise visit:
i). Why are inner transition metals at the bottom of periodic table?
two). Deviation between transition and inner transition elements.
Curt answer: I don't know exactly (lol…)
Permit me explain to you why I do not know this answer.
A full general question which all students have is about the full number of metals on the Periodic table.
Well, there is not the exact respond to this question.
Only out of the total 118 known elements, more than 78% of the elements show metallic character.
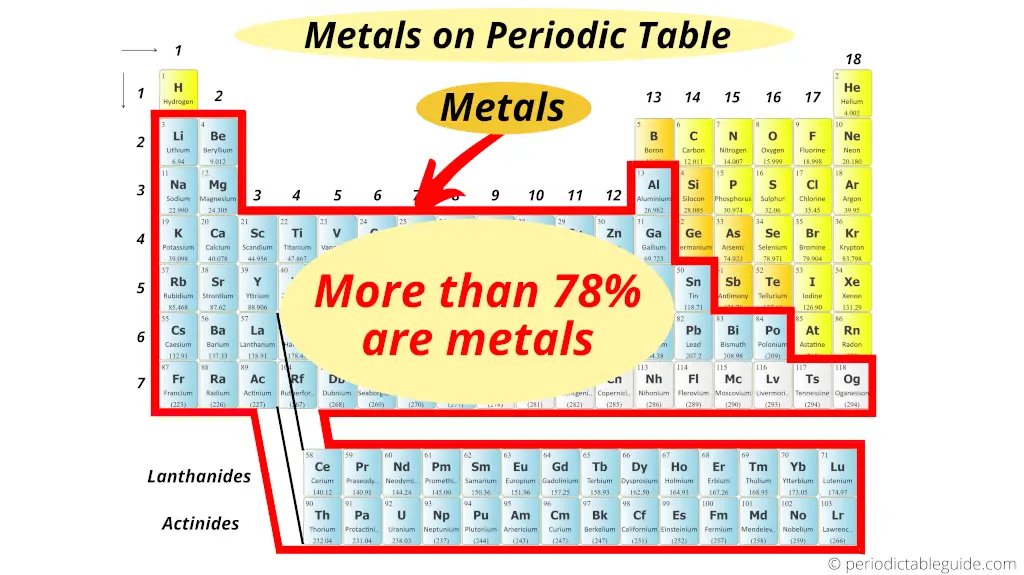
Out of the 118 elements of periodic table, approximately 94 elements are metals (merely this is non the exact number).
Reason?
According to wikipedia, this number is inexact because the boundaries between metals, nonmetals and metalloids fluctuate slightly due to lack of universally accepted definitions.
(That ways in metallurgy, the researchers may define metals on the basis of density. In physics, they may define metals on the basis of physical properties. And in chemistry, the chemists are concerned with the chemical properties of metals.)
Too the elements which are shown in grey color are however under research piece of work by the researchers (elements 109 to 118 mentioned in grey color).
These are the synthetic elements and they have a very curt half life.
This is the reason why the full number of metals are still not known.
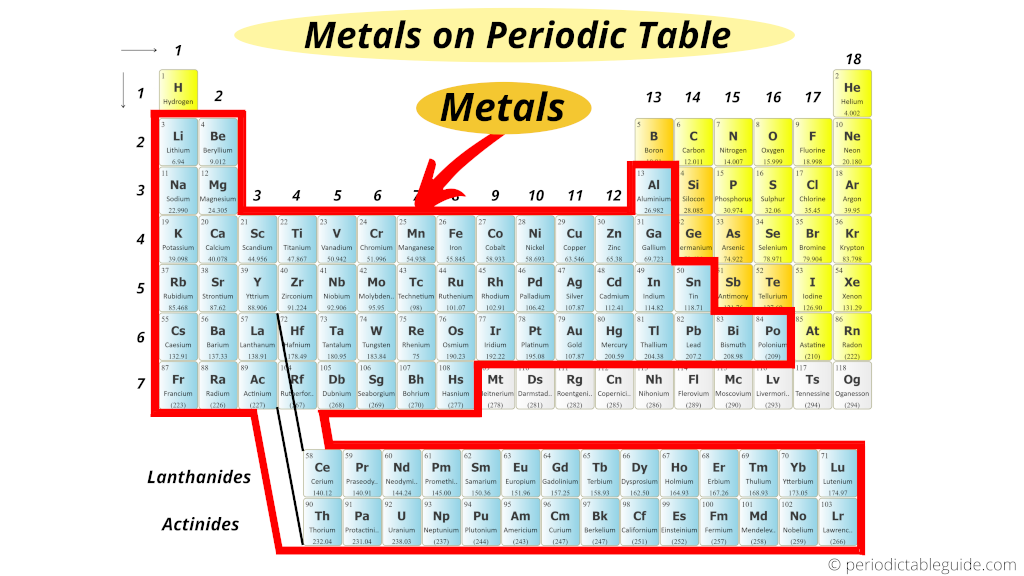
So to exist exact, if we consider the beginning 108 elements, and then there are total 84 metals present on Periodic table.
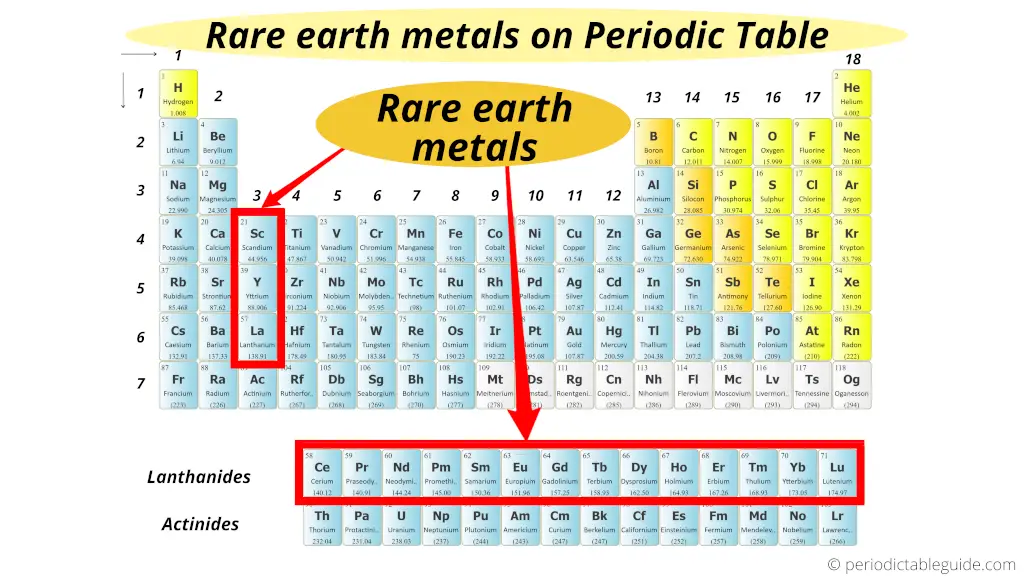
At that place are total 17 Rare Earth metals on the Periodic table. Rare Earth Metals includes all the 15 Lanthanides as well equally scandium(Sc) and yttrium (Y). So total fifteen + 2 = 17 Rare Earth metals.
Here is a consummate list of all the 17 rare globe metals.
| Diminutive number | Symbol | Proper name of element |
| 21 | Sc | Scandium |
| 39 | Y | Yttrium |
| 57 | La | Lanthanum |
| 58 | Ce | Cerium |
| 59 | Pr | Praseodymium |
| 60 | Nd | Neodymium |
| 61 | Pm | Promethium |
| 62 | Sm | Samarium |
| 63 | Eu | Europium |
| 64 | Gd | Gadolinium |
| 65 | Tb | Terbium |
| 66 | Dy | Dysprosium |
| 67 | Ho | Holmium |
| 68 | Er | Erbium |
| 69 | Tm | Thulium |
| 70 | Yb | Ytterbium |
| 71 | Lu | Lutetium |
But do you know why they are called rare?
Why Rare Earth metals are called so?
Answer: You might be thinking that rare means these elements are available in very less quantity.
Simply if you are thinking and so, and so y'all are wrong.
Rare earth metals are non actually that rare every bit the name suggests.
Even, they are available in more abundant quantities than gold.
But the fact is that they are spread evenly on the earth and it is very difficult to find these elements at one place on the earth.
Thus they are rare in the context of available resources.
First of all,
What are heavy metals?
The heavy metals are those metals which possess higher density or higher atomic mass.
Well, the definition of heavy metals may differ in metallurgy, physics equally well as chemistry.
In metallurgy, researchers define heavy metals on the basis of density.
While in physics, they may apply diminutive numbers for defining heavy metals.
Whereas in chemistry, chemists are concerned with the chemical properties of heavy metals for defining them.
So in that location is a inquiry piece of work still going on for the specific definition and classification of heavy metals.
Many researchers employ the common criteria that if the metals take the density of more than 5 1000/cm³, then they are probable to be known as heavy metals.
On the ground of this criteria, the heavy metals are shown on the Periodic table.
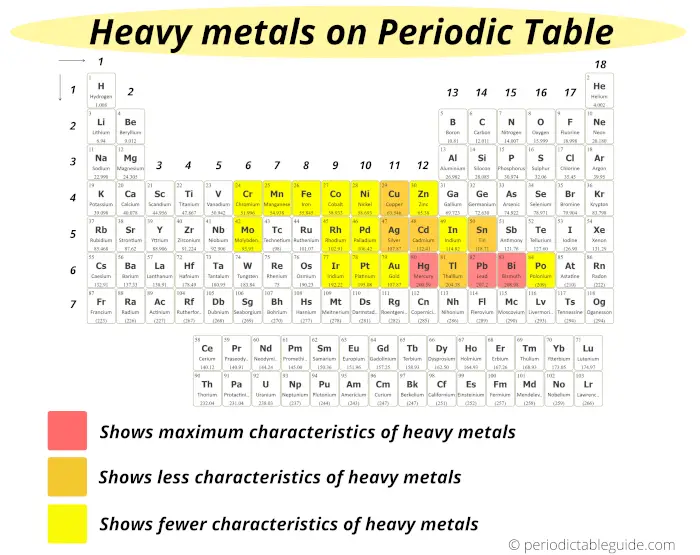
The metals which evidence maximum characteristics of heavy metals are;
- Mercury (Hg)
- Lead (Pb) and
- Bismuth (Bi)
These heavy metals are displayed on the Periodic table with red color (see above image)
The metals which show less characteristics of heavy metals are;
- Copper (Cu)
- Argent (Ag)
- Cadmium (Cd)
- Tin (Sn)
- Thallium (Tl)
And these elements are represented past orange color on the Periodic table.
Rest of the elements which are shown in xanthous color testify fewer characteristics of heavy metals.
Here I'll tell you near the most reactive metallic on the Periodic table. But earlier that, permit u.s. discuss what is a reactivity in metals?
What are reactive metals?
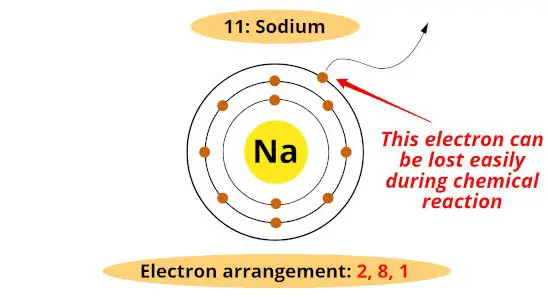
Reactive metals are those metals which evidence high tendency to lose electron/s in a chemical reaction.
For example, every bit we move downwardly the group, the diminutive size increases.
Thus the force of attraction between nucleus and outermost electrons decreases.
Considering of this, the electron will be lost easily during chemical reaction.
Hence, down the grouping, as the atomic size increases, the reactivity of metals increases.
Most reactive metallic on Periodic table
The highly reactive metals are located on the left bottom corner of the Periodic tabular array. They are the Brine metals of group 1.
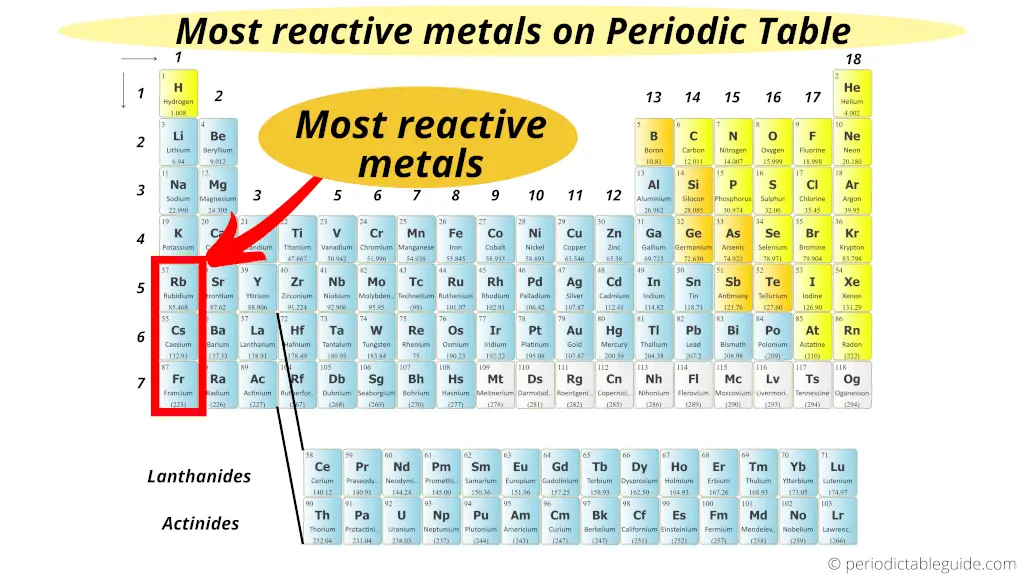
In 1st group, every bit we motility down from elevation to lesser, the reactive of metals increases.
Thus the bottom most element of group ane (i.e francium) is the most reactive metallic on the Periodic tabular array.
(Note: Francium is a laboratory made chemical element. It is bachelor in a very less quantity. And hence for whatsoever applied purposes, caesium is considered as the most reactive metal in Periodic table. Caesium has high reactivity, merely information technology is predicted that Francium would accept fifty-fifty higher reactivity than that of caesium)
Also run across: Reactivity of alkali metals with water (Explained with animation)
| Atomic number | Symbol | Proper name of element |
| iii | Li | Lithium |
| four | Be | Beryllium |
| 11 | Na | Sodium |
| 12 | Mg | Magnesium |
| 13 | Al | Aluminum |
| 19 | K | Potassium |
| 20 | Ca | Calcium |
| 21 | Sc | Scandium |
| 22 | Ti | Titanium |
| 23 | V | Vanadium |
| 24 | Cr | Chromium |
| 25 | Mn | Manganese |
| 26 | Atomic number 26 | Iron |
| 27 | Co | Cobalt |
| 28 | Ni | Nickel |
| 29 | Cu | Copper |
| 30 | Zn | Zinc |
| 31 | Ga | Gallium |
| 37 | Rb | Rubidium |
| 38 | Sr | Strontium |
| 39 | Y | Yttrium |
| forty | Zr | Zirconium |
| 41 | Nb | Niobium |
| 42 | Mo | Molybdenum |
| 43 | Tc | Technetium |
| 44 | Ru | Ruthenium |
| 45 | Rh | Rhodium |
| 46 | Pd | Palladium |
| 47 | Ag | Silver |
| 48 | Cd | Cadmium |
| 49 | In | Indium |
| 50 | Sn | Tin |
| 55 | Cs | Caesium |
| 56 | Ba | Barium |
| 57 | La | Lanthanum |
| 58 | Ce | Cerium |
| 59 | Pr | Praseodymium |
| 60 | Nd | Neodymium |
| 61 | Pm | Promethium |
| 62 | Sm | Samarium |
| 63 | Eu | Europium |
| 64 | Gd | Gadolinium |
| 65 | Tb | Terbium |
| 66 | Dy | Dysprosium |
| 67 | Ho | Holmium |
| 68 | Er | Erbium |
| 69 | Tm | Thulium |
| 70 | Yb | Ytterbium |
| 71 | Lu | Lutetium |
| 72 | Hf | Hafnium |
| 73 | Ta | Tantalum |
| 74 | Due west | Tungsten |
| 75 | Re | Rhenium |
| 76 | Bone | Osmium |
| 77 | Ir | Iridium |
| 78 | Pt | Platinum |
| 79 | Au | Gilt |
| eighty | Hg | Mercury |
| 81 | Tl | Thallium |
| 82 | Pb | Lead |
| 83 | Bi | Bismuth |
| 84 | Po | Polonium |
| 87 | Fr | Francium |
| 88 | Ra | Radium |
| 89 | Air conditioning | Actinium |
| 90 | Thursday | Thorium |
| 91 | Pa | Protactinium |
| 92 | U | Uranium |
| 93 | Np | Neptunium |
| 94 | Pu | Plutonium |
| 95 | Am | Americium |
| 96 | Cm | Curium |
| 97 | Bk | Berkelium |
| 98 | Cf | Californium |
| 99 | Es | Einsteinium |
| 100 | Fm | Fermium |
| 101 | Md | Mendelevium |
| 102 | No | Nobelium |
| 103 | Lr | Lawrencium |
| 104 | Rf | Rutherfordium |
| 105 | Db | Dubnium |
| 106 | Sg | Seaborgium |
| 107 | Bh | Bohrium |
| 108 | Hs | Hassium |
| 109 | Mt | Meitnerium |
| 110 | Ds | Darmstadtium |
| 111 | Rg | Roentgenium |
| 112 | Cn | Copernicium |
| 113 | Nh | Nihonium |
| 114 | Fl | Flerovium |
| 115 | Mc | Moscovium |
| 116 | Lv | Livermorium |
| 117 | Ts | Tennessine |
| 118 | Og | Oganesson |
Exercise you know what is the main property of metallic?
They lose electron/s during the chemical reaction.

At present just think, what should be the conditions or properties that an element should possess to make the electron lost easily.
Here are few properties necessary for piece of cake loss of electrons.
- Element should take low ionization energy
- Element should have less number of electrons in outermost orbit
- Element should take less electronegativity
If the elements possess these properties, then they will behave like metals.
Now, yous know that the elements on the left side of Periodic table have less valence electrons in their outermost orbit.
As well the diminutive size of these elements are more, which is favorable for the loss of electrons.
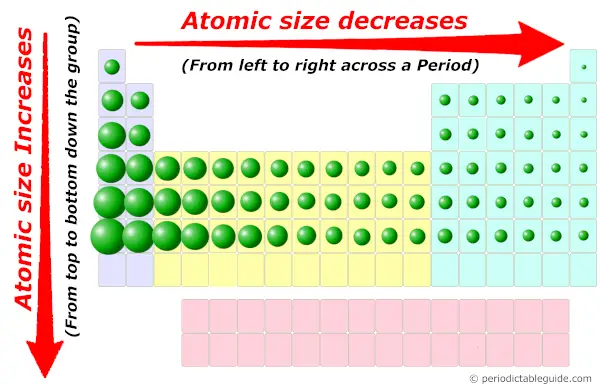
Equally they possess bigger diminutive size, information technology is easy to remove the valence electrons from the outermost orbit. So they have low ionization energy.
Also these elements located on the left side accept less electronegativity.
Permit me give yous a short introduction about electronegativity.
Electronegativity is a tendency to attract the electron pair.
I take discussed this in detailed commodity of Periodic table that electronegativity depends upon the size of an atom.
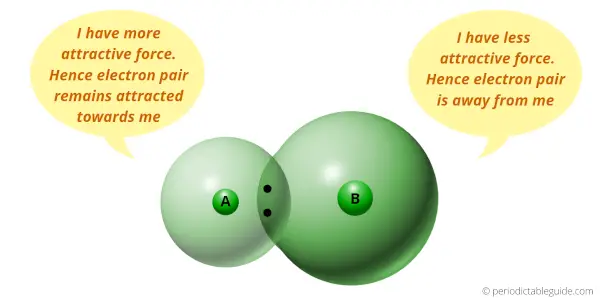
If atomic size is less, then it has more than tendency to attract the electron pair (means smaller the size, more than is the electronegativity.) And if the diminutive size is more, so information technology has less tendency to attract the electron pair (means bigger the size, lesser is the electronegativity.)
Now the elements on the left side of Periodic tabular array have more atomic size. So they will have less electronegativity.
Hence, they are more than metal in nature.
I hope, now you lot have got the respond of "Why metals are located on the left side of Periodic table?"
Hither I'll show you the physical and chemical properties of metals.
Keep reading…
Concrete properties of metal
#1 Sonorous

Metals produce ringing audio when they are stuck hard. This indicates that metals are sonorous in nature.
For instance: Ringing of bell.
#2 Solid state at room temperature

All the metals are solids at room temperature (except mercury)
They take definite shape and size.
#3 Lustrous (shiny)

Metals are Lustrous.
They have a smooth shiny surface.
#4 Malleability

Metals can be reshaped into sparse sheets on applying sufficient pressure on it. This belongings of metals is known as malleability.
#5 Ductility

Metals can be drawn into thin wires. (For instance cable wires, thin copper wires used in electric circuit used in your laptop or phone which you are holding right now, etc …)
#6 Conductivity

Metals are practiced usher of heat besides as electricity.
That means heat and electricity can easily pass through the metals.
#seven High melting point and boiling point

The metals have a loftier melting point likewise every bit boiling bespeak.
#8 Hardness
Hardness is the ability of a material to resist habiliment, tear, scratching and to resist the changes in shape.
Generally, all the metals prove these properties (except mercury)
(Note: Sodium, potassium and lithium are soft metals which can be cut with a kitchen knife.)
#ix Density

Metals have loftier density. And because of this, metals take more weight.
Chemical properties of metallic
#1 Valency
The atoms or metals have generally 1, two or iii electrons in the outermost orbit, and they lose these electrons during a chemical reaction.
#2 Corrosiveness

Some metals go corroded easily when they are exposed to moist air or water (For example: Iron)
#3 Loss of electrons

Metals have a trend to lose electrons. They donate electrons during a chemical reaction.
#4 Depression electronegative
Metals are less electronegative and so they are electropositive in nature.
#v Reactive
Some metals are chemically more reactive. They undergo chemical reactions by themselves or other elements and release energy.
For example: Potassium gets ignited automatically when information technology is exposed to water.
#6 Forms basic oxides
Metals react with oxygen and form basic oxides.
#7 Good reducing agents
Metals are reducing agents because they donate electron/due south during a chemical reaction and become oxidized.
Free Gift for y'all: Interactive Periodic Tabular array
Allow me tell you how this Interactive Periodic Tabular array volition help you lot in your studies.
1). You tin effortlessly observe every unmarried detail almost the elements from this single Interactive Periodic table.
ii). You will get the detailed information about the periodic table which will convert a newbie into pro.
three). You will also go the Hd images of the Periodic table (for Free).
Checkout Interactive Periodic table and download it's high resolution epitome now (It's FREE)
Summary
Then in the entire article, nosotros take discussed the location of Metals on the Periodic tabular array.
We saw the;
- Position of Alkali Metals,
- Position of Alkaline Globe Metals,
- Position of transition metals,
- Position of Inner transition metals,
- Position of Rare world metals,
- Position of Heavy metals too as reactive metals.
And so we discussed the complete list of all the metals of the Periodic tabular array.
I gave you the reason why the metals are located on the left side of the Periodic tabular array.
And finally we discussed the physical and chemic properties of metals.
I hope this article "Where are Metals located on the Periodic Table" has helped you in solving your queries.
Suggested Important articles for you:
- Everything you need to know almost the periodic tabular array
- Periodic table with different types of metals
- Nonmetals on the periodic table
- Metalloids on the periodic table
- Periodic tabular array labeled with metals nonmetals and metalloids
- Halogens on Periodic table
- Noble gases on periodic table
- List of elements of Periodic table
- Periodic table with electron configuration
- How are the elements arranged in the modern periodic table?
Source: https://periodictableguide.com/metals-located-on-the-periodic-table/
0 Response to "a shiny solid element also is ductile. what side of the periodic table is it likely to be found"
Post a Comment